This article is the first part of a three-part series. It explores the evolution of flowering plants and basic flower anatomy. The second article will consider the role of pollinators and the third, the cultural significance of flowers.
Before Flowers

The history of plants on Earth began long before the advent of flowering plants, the angiosperms. Before angiosperms evolved, land was dominated by early gymnosperms and expansive forests of tree ferns reaching heights of 50 feet or more. Arthropods had long since transitioned onto land, diversifying into millipedes and insects. By the time dinosaurs were established 230 million years ago, they roamed these dense, humid landscapes shaped by towering fronds, conifers, and cycads rather than flowering trees.
Early land plants first appeared in the mid‑Ordovician (about 470–458 million years ago). These pioneers were small, moss‑like organisms without vascular tissue, so they clung to moist environments and reproduced using spores rather than seeds. Green algae (charophyte algae), the aquatic freshwater ancestors that gave rise to these pioneers, were the ultimate predecessor of all land plants. Flowering plants were and are the latest evolutionary development, but they incorporate innovations achieved across millions of years which allowed plants to survive and thrive on land. Moving onto land required solving three major problems: drying out, standing up, and reproducing without submersion in water. To prevent drying out, plants evolved a waxy outer coating (the cuticle) and tiny adjustable pores (stomata) that let in carbon dioxide for photosynthesis while limiting water loss. To stand upright and transport fluids, they developed supportive tissues stiffened with lignin (a natural glue), along with “plumbing” — xylem and phloem — to transport water, minerals, and sugars throughout the plant. Early root-like structures (rhizoids), and later roots, enabled them to anchor into soil and tap into nutrients and water below its surface. Durable spores powered the first wave of land colonization. Much later, seed plants made a bigger leap: seeds that protected and nourished the embryo and freed reproduction from standing water.
Some of the plants that evolved before flowering plants survive today in niche or well-defined environments, but flowering plants now dominate the biome. Many grow fast, propagate with insane speed, and have perfected specializations that enable them to seemingly grow anywhere. Angiosperms feed us in countless ways—their structures, seeds, and fruits form the core of our diet—and they sustain many other animals as well. Deer and rabbits rely heavily on them because flowering plants dominate their habitats and offer abundant, nutritious new growth. They favor tender shoots and young leaves, though many angiosperms have evolved defenses — such as tough leaves (hollies and oaks), bitter compounds (milkweeds and nightshades), or spines (cacti) — that limit browsing.
Flowers predated humans by over a hundred million years. Long before flowers evolved, early pollinators fed on the nutritious, sugary pollination drops produced by ancient gymnosperms. Flowers first appeared roughly 140 million years ago—the age of the oldest generally accepted angiosperm fossils—during the Early Cretaceous, a time dominated by dinosaurs, early birds, small shrew‑like mammals, and diverse insects such as beetles, flies, and wasps (bees evolved from predatory wasps and appeared slightly later in the Cretaceous). Fossil evidence shows that beetles and scorpionflies were already pollinating non‑flowering seed plants nearly 300 million years ago. In contrast, modern humans (Homo sapiens) did not evolve in Africa until about 200,000 to 300,000 years ago.
Seed plants, such as conifers and ginkgoes (yes, they are still with us), appeared between 350 and 310 million years ago. These early gymnosperms were the first plants to use pollen. These innovations — seeds and pollen — moved reproduction from water to land. Early land plants, from tiny moss‑like forms to the first ferns, used spores for dispersal but their sperm still had to swim through a film of water to reach an egg—a strategy many of their descendants still use today. In ferns, sperm typically fertilize an egg on the same gametophyte, whereas in mosses they usually swim to the eggs of a different plant. True ferns never evolved seeds, but several no longer existing fern‑like lineages did. These “seed ferns” weren’t real ferns; however, they produced true seeds and helped give rise to early seed plants. Though those groups eventually disappeared, their seed‑bearing descendants led to gymnosperms and, much later, flowering plants.
Gymnosperms: Pollen and Seeds

Gymnosperms are mostly evergreen and produce exposed, or “naked,” seeds on leaves or cones, with pollen produced in male cones and ovules in female cones. Seeds offer major advantages over spores: they can disperse over long distances without the need for water, they protect an embryo supplied with stored food, and they can remain dormant for extended periods. Spores, by contrast, are single cells with no food reserve, minimal protection, and an immediate dependence on moisture.
Gymnosperm pollination occurs primarily by wind, though fossils show pollen and reproductive tissues associated with insects. Sticky and sugary pollen droplets, nutritious fluids that serve as an evolutionary analogue to nectar, trap airborne pollen and suck it into the ovule. Some insects feed on it and, in the process, transfer pollen. Conifers and ginkgoes are wind‑pollinated, while cycads and many gnetophytes—a small but distinct gymnosperm lineage—rely on insects. Wind pollination demands the production of large quantities of pollen.
While seed dispersal in gymnosperms is largely wind‑driven, birds and mammals contribute in some species. Squirrels—and less frequently raccoons—eat ginkgo seeds, especially once the fleshy outer layer has decayed enough to expose the nut inside, but the large, strong‑smelling sarcotesta (the outer seed coat) likely evolved to attract much larger, now‑extinct dispersers. Although the ginkgo appears to produce a fruit, this structure is actually a naked seed with a fleshy outer coat.
Seeds enabled early gymnosperm ancestors to colonize land, but angiosperms revolutionized this strategy by enclosing seeds within fruits— structures that protect developing embryos and entice animals to facilitate broad dispersal. Emerging from an extinct, gymnosperm-like lineage, flowering plants did not arise directly from cones. Instead, genetic studies suggest their floral structures evolved through modified or tweaked genetic coding in a common ancestor. These adaptations, paired with more efficient vascular systems and a shift toward pollination by animals, allowed angiosperms to surpass the efficacy of wind-pollination.
The essential distinction between gymnosperms and angiosperms lies in the names themselves: the term gymnosperm derives from Greek gymnos (“naked”) and sperma (“seed”), while the term angiosperm comes from angeion (“vessel”) and sperma, referring to seeds enclosed within a fruit.
The Emergence of Flowering Plants
When angiosperms emerged, Earth’s atmosphere was very different from today—a topic that remains an active area of scientific research. Current evidence suggests that CO₂ levels were far higher, warming the planet, while earlier land plants had already raised oxygen close to modern levels. Scientists hypothesize that as flowering plants spread, they increased atmospheric moisture and reshaped fire and wetland patterns, indirectly influencing the cycling of carbon and oxygen—the ongoing exchange between life, the atmosphere, and Earth’s surface.
Evolution practices both randomness and predictability. Random mutations, genetic recombination, shifts driven by external forces, and changing environments introduce unpredictability, while natural selection filters these variations, favoring traits that improve survival and reproduction. Such traits tend to persist. Sometimes different lineages arrive at similar solutions independently—a pattern known as convergent evolution, as seen in the wings of birds, bats, and insects. Plants do the same thing. In short, different life forms can respond to similar environments by developing similar traits. For instance, climbing plants across unrelated families independently evolved structures such as tendrils to access sunlight without developing a thick trunk. When we consider flower shapes, however, coevolution is the central story: plants and pollinators shaping one another because each depends on the other. Evolution is a dance, though the partners are unaware of their steps. And while evolution can appear directed, it is not a climb up a ladder; it produces immense diversity, and natural selection simply preserves what works in a given context. Although the maximum complexity of life has increased over deep time, evolution does not inherently drive organisms toward greater complexity. It favors whatever traits allow organisms to survive and reproduce more effectively in their current environment.
Flower Form and Function
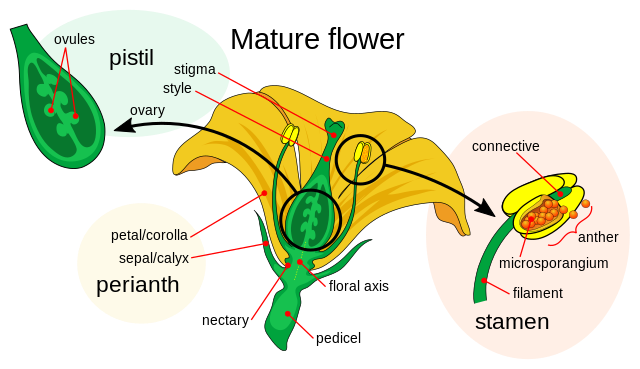
All angiosperms produce flowers—this is the defining feature of the group. A flower is an extraordinary evolutionary innovation, built to facilitate sexual reproduction. Sexual reproduction generates genetic diversity, helping lineages avoid repeating maladaptive traits and driving the coevolution of plants and their pollinators.
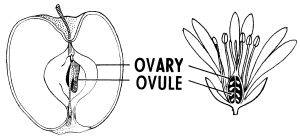
Flowering plants utilize a vast array of morphological and genetic adaptations to achieve their core reproductive goals, typically arranging their organs in concentric rings known as whorls. The central female structure, the pistil (or carpel), consists of a pollen-receptive stigma at the top, a connecting style, and an ovary at the base. Within the ovary are ovules, which house the female gametes; once fertilized by pollen growing through the style, these ovules mature into seeds. Seeds are alive with a tiny embryo inside and on pause. Under unfavorable conditions, and over time, they can die. This is because they slowly use up their stored food. A seed is botanically defined as a matured, fertilized ovule, so the term “unfertilized seed” is technically incorrect, because unfertilized structures remain ovules. However, some plants, such as dandelions and mangos, can produce cloned embryos through a process known as apomixis. Fertilization is bypassed but not the ovule. Following successful fertilization, the ovary develops into a fruit, the final size of which is independent of the original flower’s size. Fruit shapes often evolve to attract animals that disperse seeds internally—a process known as endozoochory—with the seeds protected during passage through the animal’s digestive tract. In some species, fruits incorporate additional floral or stem tissues beyond the ovary itself.
The male structures are the stamens, each consisting of a filament topped by a pollen-producing anther which it supports. The anther releases pollen — sometimes thousands of grains — for fertilization. The connective is the sterile tissue in the anther that connects the two pollen sacs. The microsporangium (typically four per anther) is one of the tiny pollen chambers where cells undergo meiosis, a two‑step division that halves and reshuffles the DNA to produce genetically unique microspores. Each microspore develops into a pollen grain. In some species, the pollen grain already carries its two sperm cells when it’s released (as in grasses and sunflowers). In others, the pollen grain carries a generative cell instead—a small cell tucked inside the pollen that will later split into two sperm as the pollen tube grows toward the ovule (as in lilies, tulips, and roses). Two sperm cells are necessary because flowering plants use double fertilization: one sperm makes the embryo and the other makes the endosperm that feeds it. Both the embryo and the endosperm (embryo food) end up in the seed. By contrast, gymnosperms provide nourishment for the embryo with nutritive tissue that develops before fertilization.
A single flower can bear multiple stamens and multiple carpels, or it may lack one sexual function entirely, influencing how widely its genes are exchanged. Some flowers are even sterile, producing neither functional stamens nor carpels. In many species (for example, bigleaf hydrangea, poinsettia, and Queen Anne’s lace) these sterile flowers enhance pollinator attraction to nearby fertile flowers, indirectly increasing opportunities for cross‑pollination and genetic mixing.
The perianth is comprised of petals and sepals which surround the reproductive organs when present. Petals are usually colorful, while sepals are typically green, though both can be reduced or absent in some species. When petals and sepals are indistinguishable, the segments are called tepals. This is common among monocots which are discussed in detail in a later section. The calyx encompasses the flower’s sepals, which shelter a bud, and the corolla refers to the whorl of petals that surround the reproductive parts. The floral axis is the central stem to which floral parts connect, while the pedicel is a slender stalk that supports a flower. Finally, the nectary is the gland that produces a sugary liquid to attract and reward pollinators.
The Appearance of the First Flower

When 19th‑century scientists began examining the planet’s geology, flora, and fauna through an evolutionary lens and with a growing fossil record, flowers quickly became a compelling puzzle. They are ubiquitous, diverse, and clearly central to plant reproduction. It is natural to wonder when they first evolved, why there are so many different kinds, and what purpose they serve. Because of the flower’s apparent “sudden” appearance and rapid differentiation, evolutionist Charles Darwin (1809-1882) called the appearance of flowers an “abominable mystery.” He saw evolution as a gradual journey, and he was perplexed by the relative rapidity of the spread of angiosperms, attested to by their abrupt appearance in the fossil record. (In an 1879 letter to botanist Joseph Hooker he wrote: “The rapid development as far as we can judge of all the higher plants within recent geological times is an abominable mystery.” vol. 27, 1879, The Correspondence of Charles Darwin, Cambridge University Press, 2020). Darwin suggested that their proliferation was the result of coevolution between flowering plants and insects. He explored this in his book The Various Contrivances by Which Orchids Are Fertilized by Insects (1877, 2nd ed.). He declared that the visits of insects are indispensable and flowers have adapted to secure their aid. The idea was not entirely new. The concept itself had earlier roots. In 1793, Christian Konrad Sprengel (1750–1816) published pioneering observations on pollination biology, recognizing the role of insects in transferring pollen. Even earlier, in the 17th century, Nehemiah Grew (1641–1712) and Rudolph Jakob Camerarius (1665–1721) established that pollen functions as the male reproductive element in plants, analogous to sperm in animals.
The quest for the first flower continues. It reminds us that flowering plants did not appear suddenly or without precedent. Scientists now view angiosperms as a logical evolutionary development from earlier seed plants, and their origins may be older than the widely cited 140‑million‑year mark. Because flowers preserve poorly, the fossil record is sparse. The oldest widely-accepted fossil flower is Montsechia vidalii, a small aquatic plant from about 130 million years ago, slightly older than Archaefructus at roughly 125 million years. However, molecular clock studies (for an explanation of how a molecular clock works, watch the video Molecular Clocks by Lumina Learning) suggest that angiosperms may have originated in the Jurassic—or even the Triassic—long before their first clear fossils. The absence of older fossils may reflect early angiosperms living in environments unfavorable to fossilization, being extremely small and fragile, or simply being very rare.
What did the first flowers look like? It’s difficult to say, but there are theories. Some suggest they were parts of small weedy plants opportunistically growing in the understory of gymnosperm-dominated landscapes, or perhaps they developed in an aquatic environment and sort of resembled a water lily. Whatever its appearance, there is strong evidence that all flowering plants evolved from a common ancestor and diversified quickly across the supercontinents of Laurasia and Gondwana.
Sixty-six million years ago the Cretaceous Period ended with the impact of a huge asteroid, resulting in the mass extinction of roughly 77% of all species. Plants were impacted less than animals; nonetheless, because of the atmospheric response, many plant species expired. There were a number of reasons for their destruction: acid rain, lack of sunlight, and expansive firestorms ignited by superheated material hurled into space and then returning to Earth, which heated the atmosphere to extreme temperatures. The result was a planet open to the spread of the endlessly adaptable angiosperm. From trees, to grasses, to shrubs, to ornamental flowers, to weedy plants, and our food crops – today angiosperms are pretty much everywhere. Noting the vast expanse of time reminds us of how much “room” there has been for plants and other forms of life to evolve. This long evolution, indicated above, is measured in geological time by using definitive changes in rock layers and by fossils which mark the boundaries between units and is expressed in eons, eras, periods, epochs, and ages.
Again, what did the first flowers look like? The debate is lively. So why bother? Well, beyond the virtue of curiosity, knowing will tell us how an important part of our world began and will reveal the factors that shaped it. Adding this knowledge to what we already know about angiosperms can guide breeders, bioengineers, and horticulturists as they develop hardier, more resilient, and more adaptable flowering plants.


Amborella trichopoda, a shrub found only on the island of New Caledonia in the southwest Pacific Ocean, is the closest living relative of all other flowering plants and provides important clues about early angiosperm evolution. Although not a model of the first flower itself, Amborella retains several ancestral features—such as spirally arranged tepals, separate or unfused reproductive organs, and primitive xylem—that help illuminate early floral traits. Magnolia‑like flowers historically served as a model for early angiosperms, with numerous spirally inserted tepals, stamens, and separate carpels on an elongated receptacle and were relatively large, though magnolias diverged later than Amborella. Early angiosperms likely emerged in warm, moist understories, similar to the modern habitat of Amborella trichopoda.
Using data from the eFLOWER Initiative (a prestigious international collaboration) botanists analyzed nearly 800 living angiosperms to reconstruct the ancestral flower. Instead of finding a fossil, they did evolutionary reverse-engineering, mapping the traits of these living species onto a enormous DNA family tree. They then employed statistical models to calculate the most likely model for the first flower. The result is sometimes compared loosely to magnoliid or water lily flowers, but Initiative scientists note that any resemblance is merely superficial. While fossil evidence from the Early Cretaceous shows a mix of structures, the eFLOWER Initiative reconstructed the ancestral flower as bisexual and radially symmetrical. Because the reconstruction is not based on a fossil, inferring size is uncertain but — based on early lineages — small is often inferred. The primary reconstruction can readily be found on the eFLOWER Initiative’s homepage and a high quality image is included in the 2017 Nature Communications article noted below.
The eFLOWER Initiative (see the website specifically for the eFLOWER Project and Diversification of Angiosperms (Sauquet lab)) challenges the long‑held idea that the first flowers resembled magnolias with their organs arranged in a loose spiral. Instead, eFLOWER proposes that early flowers were mostly organized in tidy, circular layers, each made of three repeating parts. The one exception is the carpels, where the statistical support is weaker—leaving open the possibility that this inner layer might have been spiral. That uncertainty is what fuels the so‑called “hybrid” controversy. Some researchers remain skeptical because the reconstructed flower doesn’t closely resemble any living species, depends heavily on statistical modeling, and has limited support for certain traits—especially how the floral organs were arranged as they grew inside the bud. For a pertinent discussion, which first formally presented the model, see the 2017 article in Nature Communications “The Ancestral Flower of Angiosperms and Its Early Diversification.” For a look at responses, see the 2018 article “Unbuttoning the Ancestral Flower” in Trends in Plant Science.” It challenges the eFLOWER conclusion that the first flower had circular parts (whorls) rather than spiral ones and questions the statistical reasoning behind the reconstruction. Most recently, the 2024 Nature article “Phylogenomics and the Rise of Angiosperms” uses massive DNA datasets to explain how and when angiosperms conquered the planet, complementing eFLOWER’s morphological focus. It does not challenge the eFLOWER model; instead, it confirms the evolutionary roadmap and branching structure used in its mathematical analyses. For an update on where the effort stood in 2024, see The eFLOWER project: what have we done, where are we, and where are we heading now?
Did dinosaurs play a role in angiosperm emergence? Dinosaurs roamed Earth for nearly 100 million years before the first angiosperms appeared in the Early Cretaceous, and the two groups coexisted for roughly 70 million years. Scientists have long wondered how they influenced one another. The consensus is that dinosaurs did not cause flowering plants to evolve, but many herbivorous dinosaurs later incorporated angiosperms into their diets. Some researchers have proposed that heavy dinosaur browsing on gymnosperms may have opened up disturbed habitats—much like fire or storm damage—creating opportunities for fast‑growing angiosperms to colonize. However, the major drivers of angiosperm spread were their own reproductive innovations and their partnerships with pollinating insects.
Divergence: Monocots, Eudicots, and Basal Angiosperms
According to data from the Royal Botanic Gardens, Kew and Encyclopaedia Britannica, there are roughly 352,000 formally described species of living angiosperms. While estimates often range from 250,000 to 400,000, these flowering plants now represent nearly 80% of all land plant species, making them the most ecologically dominant group on Earth. The differences in counts can be explained as a consequence of the same plants being named differently by scientists across the globe, plus the uncertainty of when a variation becomes a species, and the complexity of groups that hybridize readily. These figures encompass diverse aquatic lineages as well, including water lilies (freshwater), pondweeds (almost entirely freshwater but some species tolerate brackish water), and seagrasses (exclusively saltwater). Botanists continue to describe roughly 2,000 new living species annually, a pace driven by DNA sequencing, the re-examination of preserved plant specimen collections, and the exploration of global biodiversity hotspots.
Early in flowering‑plant evolution, a few small lineages split off long before the rise of the major groups we see today. For botanists, a lineage is simply a branch of a plant’s family tree: as evolution proceeds, one branch can split into two, and each becomes its own lineage. DNA evidence shows that the ANA grade —Amborellales (Amborella), Nymphaeales (water-lily group) and Austrobaileyales (star-anise group) —represents the very first three branches. Amborella sits at the base as the earliest sister to all other living angiosperms. These early splits occurred well before the burst of diversification that produced the large modern lineages known as the core angiosperms Mesangiospermae). Eudicots and monocots are the largest groups in the core, but magnoliids, Chloranthales, and Ceratophyllales are also part of it. Eudicots comprise roughly 75% of all angiosperms. This framework comes from the work of the Angiosperm Phylogeny Group (APG), whose DNA‑based classification clarified these deep evolutionary relationships. These distinctions form the basis of what botanists loosely refer to as the basal angiosperms.
The original monocot–dicot distinction, introduced by John Ray in 1682, was based solely on morphology: monocots have one cotyledon (embryonic leaf) and dicots have two. The terms come from the Greek cotyledon, meaning “small cup,” referring to the seed leaves inside the embryo. While the cotyledons are the namesake, pollen aperture structure is the most reliable morphological “marker” for eudicots (they have three pores/furrows, known as triaperturate, while monocots and basal angiosperms typically have one). Modern classification still considers traits such as seed‑leaf number, flower‑part arrangement, leaf venation, vascular‑bundle patterns, root systems, and pollen‑aperture structure, but DNA evidence was the decisive factor in revising the system. DNA-based classification is the work of the Angiosperm Phylogeny Group (APG). Its work led to the removal of several groups from the old “dicot” category because DNA showed they weren’t actually part of the true dicot family tree. Nonetheless, most former dicots were reassigned to the eudicots — eu meaning “true” — while a few ancient lineages were separated out as basal angiosperms.
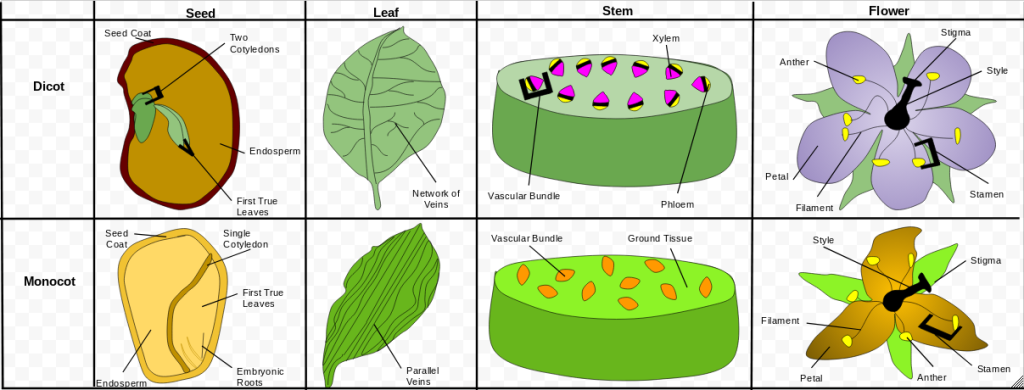
Floral characteristics can help distinguish monocots from eudicots, though they are not definitive on their own. Monocots typically have floral parts in multiples of three (for example, 3 or 6 petals), while eudicots usually show parts in multiples of 4 or 5. Radial floral symmetry is common in basal angiosperms and remains the standard across most monocots and eudicots. However, many major modern lineages have independently evolved bilateral flowers to accommodate specific pollinators and this is most common among eudicots. Monocot perianths are often undifferentiated into distinct sepals and petals, whereas eudicots more commonly have a clearly differentiated calyx and corolla. Radial symmetry can be visualized as a bicycle wheel with spokes radiating from a hub; bilateral symmetry resembles a butterfly folded along its midline, with mirrored halves. These floral “shape” traits reflect evolutionary pressures from pollinators and environments rather than serving as strict diagnostic criteria for classification. In fact, the fundamental distinction among monocots, eudicots, and basal angiosperms lies in their evolutionary lineages as revealed by molecular and pollen‑structure evidence, rather than by any single visible trait.
In conclusion, the three major angiosperm lineages emerged as early flowering plants diverged into distinct evolutionary paths, each following different genetic and developmental trajectories. In a world undergoing rapid environmental change, even small genetic shifts produced large structural differences, allowing multiple strategies to flourish simultaneously. As noted above, the earliest branches became the basal angiosperms, many of which today persist in shaded, wet, and relatively stable habitats. Later divergences produced the monocots, characterized by streamlined architecture and fibrous root systems, and the eudicots, whose highly adaptable developmental toolkit enabled them to radiate into an enormous range of environments. Examples include water lilies among basal angiosperms, grasses and sedges among monocots, and roses, sunflowers, and tomatoes among eudicots. Monocots typically form shallow, fibrous root systems, whereas eudicots commonly develop deeper taproot systems. Over more than 130 million years, these lineages diversified dramatically, leaving a legacy of three deep evolutionary trajectories that molecular evidence now clearly reveals.
Complete and Incomplete, Perfect and Imperfect
Angiosperm flowers can be classified by the presence of their major floral parts and by whether they contain both reproductive organs. A flower with sepals, petals, stamens, and pistils is complete; one missing any of these structures is incomplete. Completeness, however, has no bearing on pollination strategy. Flowers may also be perfect or imperfect: perfect flowers contain both stamens and pistils and can self‑ or cross‑pollinate, while imperfect flowers lack one sex and therefore require cross‑pollination. While perfect flowers can self‑pollinate, many have evolved mechanisms that reduce or prevent this (for example self‑incompatibility, timing differences, spatial separation). Monoecious species bear separate male and female flowers on the same plant (corn, squash), whereas dioecious species place them on different plants (holly, spinach), ensuring cross‑pollination and promoting genetic diversity. These structural categories sometimes correlate with floral form: many perfect and complete flowers are large and showy, while many imperfect or incomplete flowers—especially those adapted for wind pollination—tend to be smaller and less conspicuous. These are broad patterns rather than strict rules.
A flower has a set of tasks it must perform in its environment — among these are attracting pollinators, positioning and protecting reproductive organs for efficient pollen transfer, releasing scent or producing nectar in a timely manner, and reducing self-pollination when cross-pollination is more advantageous. Flower shape evolves to meet these tasks, and is not simply a consequence of being perfect, imperfect, complete, or incomplete. The next article in this series will remember these distinctions and explore the function or purpose of flower shape in detail.
Featured Photo: The rose (Rosa) is a classic eudicot, characterized by netted leaf venation and vascular bundles arranged in a ring. Its radially symmetric flowers feature distinct whorls—typically in multiples of five—with numerous stamens and unfused carpels. These organs sit within a hypanthium formed by the fusion of sepal, petal, and stamen bases. This cup-like structure surrounds the ovary without fusing to it, eventually ripening into a rose hip. This accessory fruit encloses multiple achenes (true simple fruits), each containing a single seed. Portland International Rose Test Garden. Photo: Vulturesong. Wikimedia Commons. CCO 1.0 Universal
Sources
Angiosperm. By Martin Huldrych Zimmermann. (2026). Encyclopaedia Britannica.
Digital Atlas of Ancient Life. Paleontological Research Institution. A truly outstanding resource: thorough explanations with wonderful illustrations.
The eFLOWER Initiative: A Framework for Understanding the Evolution and Diversification of Flowers. University of Vienna. Among the key questions the work addresses: “What were the first flowers like in deep nodes of the angiosperm tree?” On the homepage is an image of the proposed ancestral flower. It’s collaborators advance knowledge by compiling and standardizing floral trait data—creating new observations, gathering information from the literature, and organizing it into a shared database for large‑scale analyses. It is an international collaboration. For the eFLOWER Project specifically see: Diversification of Angiosperms (Sauquet lab) and eFLOWER
“How flowering plants beat bloom-free gymnosperms to world dominance.” (2022, November 12). New Scientist, 256(3412), 44–47.
Episode 57: The Evolution of Flowering Plants (Angiosperms). The Common Descent Podcast. With then doctoral student Aly Baumgartner, now Dr. Aly Baumgartner, Paleontology Collections Manager and Paleobotanist at Sternberg Museum of Natural History, Hays, Kansas, Fort Hays State University. See also the video When Plants Ruled the World in the series Evolution Soup.
Herbaceous Landscape Plants, chapter 15 in Virginia Cooperative Extension Gardener Handbook, Virginia Cooperative Extension. Useful practical advice for gardeners. See other chapters as appropriate because angiosperms are not all herbaceous.
The Science of Plants: Understanding Plants and How They Grow. Edited by Emily Tepe. University of Minnesota Libraries. Freely downloadable textbook. CC BY 4.0
Wikipedia. The Plants Portal. Gathers selected articles, illustrations, and categories. This is a great place to browse botany topics. To complement this article, see especially: Flowering Plant, Flower, and Evolutionary History of Plants.
Barrett, P. M., & Willis, K. J. (2001). “Did dinosaurs invent flowers? Dinosaur–angiosperm coevolution revisited.” Biological Journal of the Linnean Society, 74(4), 519–557.
Brusatte, S. L., Benton, M. J., Lloyd, G. T., Ruta, M., & Wang, S. C. (2008). “Dinosaurs and the Cretaceous Terrestrial Revolution.” Proceedings of the Royal Society B: Biological Sciences, 275(1650), 2483–2490.
Butler, R. J., Barrett, P. M., Kenrick, P., & Penn, M. G. (2009). “Diversity patterns amongst herbivorous dinosaurs and plants during the Cretaceous: Implications for hypotheses of dinosaur–angiosperm coevolution.” Journal of Evolutionary Biology, 22(3), 446–459.
De-Paula, Orland C., et al. “Unbuttoning the Ancestral Flower of Angiosperms.” (2018). Trends in Plant Science 23(7), 551-554.
Sauquet, H., von Balthazar, M., Magallón, S., Doyle, J. A., Endress, P. K., Bailes, E. J., Barroso de Morais, E., Bull-Hereñu, K., Carrive, L., Chartier, M., Chomicki, G., Coiro, M., Cornette, R., El Ottra, J. H. L., Epicoco, C., Foster, C. S. P., Jabbour, F., Haevermans, A., Haevermans, T., … Schönenberger, J. (2017). “The ancestral flower of angiosperms and its early diversification.” Nature Communications, 8, Article 16047.
Sauquet, H., Lopez-Martinez, A., von Balthazar, M., Chartier, M., Herting, J., Ramirez-Barahona, S., Magallón, S., & Schönenberger, J. (2024). The eFLOWER project: what have we done, where are we, and where are we heading now? In XX International Botanical Congress IBC 2024, Spain. Book of Abstracts. Oral presentations (pp. 23).
Zuntini, A.R., Carruthers, T., Maurin, O., et al. “Phylogenomics and The Rise of Angiosperms/” (2024). Nature 629, 843-850.
For simple introductions to floral morphology, see also the straightforward: Parts of a Flower (University of Florida/IFAS Extension) and Reproductive Plant Parts (Oregon State University Extension).